What is the risk of developing myocarditis or pericarditis following a SARS-CoV-2 mRNA based vaccination?
What the research says:
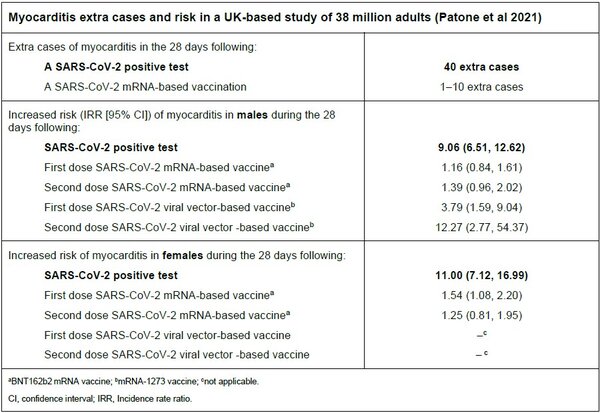
The rates of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the tissue surrounding the heart – the pericardium) are higher following COVID-19 illness than following mRNA-based SARS-CoV-2 vaccination overall, and among both males and females (Patone et al 2021). There appears to be some increase in the risk of myocarditis following mRNA-based SARS-CoV-2 vaccination, particularly among males following the second dose of the Moderna mRNA-based SARS-CoV-2 vaccine (mRNA-1273) (Patone et al 2021).
However, the benefits of mRNA vaccines against SARS-CoV-2 greatly outweigh the risk of myocarditis and pericarditis (WHO 2021). CDC recommend that patients who experience myocarditis or pericarditis following the first dose of an mRNA vaccine should not receive a second dose of another mRNA-based SARS-CoV-2 vaccine (CDC 2022).
Symptoms of myocarditis and pericarditis typically occur within a few days of vaccination with mRNA vaccines and may include new and persisting chest pain, shortness of breath, racing/pounding heartbeat. The symptoms are usually mild and self-limiting and most people recover completely with conservative treatment (non-steroidal anti-inflammatory drugs [NSAIDs], rest) with no lasting symptoms.
What this means for your clinical practice:
- Continue to offer mRNA vaccines per National guidelines, including to those patients with a history of myocarditis/pericarditis unrelated to mRNA SARS-CoV-2 vaccination if the episode has resolved completely and there is no evidence of ongoing heart inflammation or sequelae
- Where possible, young adults aged 12 to 29 years for whom an mRNA vaccine is to be offered should preferentially receive the Pfizer-BioNTech mRNA vaccine, especially for the second dose as per the Canadian National Advisory Committee on Immunization (2021)
- Be vigilant for emergent symptoms suggestive of myocarditis or pericarditis such as persisting chest pain, shortness of breath, racing/pounding heartbeat, especially in younger males
With grateful thanks to Dr Osman Yusuf (Chief Consultant of the Allergy & Asthma Institute, Pakistan) for and on behalf of the IPCRG practice driven answers review group.
Useful links and supporting references:
Canadian National Advisory Committee on Immunization. Summary of the National Advisory Committee on Immunization (NACI) rapid response of December 3, 2021. Available at: https://www.canada.ca/content/dam/phac-aspc/documents/services/immunization/national-advisory-committee-on-immunization-naci/rapid-response-recommendation-use-covid-19-vaccines-individuals-aged-12-years-older-myocarditis-pericarditis-reported-following-mrna-vaccines/summary.pdf. Accessed April 2022.
CDC. Vaccines & Immunizations. Interim Clinical Considerations (January 2022). Available at: https://www.cdc.gov/vaccines/covid-19/clinical-considerations/covid-19-vaccines-us.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fvaccines%2Fcovid-19%2Finfo-by-product%2Fclinical-considerations.html. Accessed February 2022.
Patone M, et al. Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection. Nature Medicine 2021. Available at: https://www.nature.com/articles/s41591-021-01630-0. Accessed February 2022.
WHO. COVID-19 subcommittee of the WHO Global Advisory Committee on Vaccine Safety (GACVS): updated guidance regarding myocarditis and pericarditis reported with COVID-19 mRNA vaccines. July 2021. Available at: https://www.who.int/news/item/09-07-2021-gacvs-guidance-myocarditis-pericarditis-covid-19-mrna-vaccines. Accessed February 2022.
Resource information
- COVID-19
- Respiratory Infection
- Vaccination